Inquiry
Integrating human organ models on a single microfluidic platform to study systemic physiology and drug responses.
Multi-organ-on-a-Chip (MooC) is an integrated microphysiological platform that interconnects multiple functional human 3D tissue modules through a precisely controlled microfluidic network to recapitulate systemic human physiology. By combining advanced microfabrication with human-relevant 3D tissue engineering, MooC functions as a Body-on-a-Chip, enabling dynamic simulation of absorption, distribution, metabolism, and excretion (ADME), as well as organ–organ signaling and regulatory feedback loops that govern whole-body responses.
Multi-Organ-on-a-Chip systems enable kinetic modeling of how biological effects can ripple across multiple organs over time to induce changes in organ function/systemic physiology, bridging the gap between reductionist cell cultures and live animals in a mechanistically informed manner.
Why Multi-organ-on-a-Chip?
The "productivity gap" in drug development stems from the structural failure of traditional models to capture systemic human biology.
- The Species Barrier: Metabolic capabilities differ between animals and humans ("metabolism blind spots") leading to "clinical attrition."
- Loss of Systemic Dimension: Existing organ-on-a-chip models represent isolated "biological islands." Isolated chips cannot model organ crosstalk/endocrine/metabolic feedback loops involved in maintaining whole-body homeostasis.
- Static vs. Dynamic: Traditional in vitro cultures (like Transwells) involve large liquid volumes that dilute signaling factors and lack the mechanical shear stress required for realistic tissue maturation.
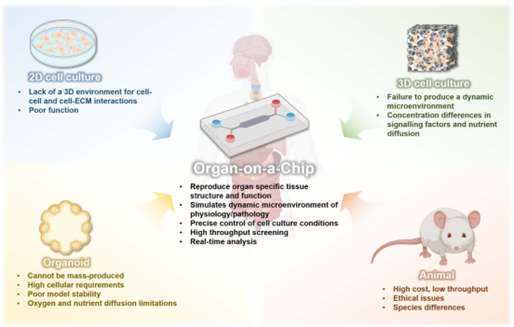 Fig. 1. Advantages of organ-on-a-chip over other models (Chen C, et al., 2025).
Fig. 1. Advantages of organ-on-a-chip over other models (Chen C, et al., 2025).
Platform Architecture and Design
Our MooC platform is built on standardized, scalable engineering principles designed to maximize physiological fidelity, reproducibility, and translational relevance.
Modular "Plug-and-Play" Integration
We employ a modular architecture that allows different organ modules (e.g., Liver, Kidney, Lung) to mature independently in their optimized microenvironments before systemic coupling via a microfluidic motherboard. Once established, organs can be connected to the microfluidic "motherboard" to introduce systemic flow.
Vascularized Barrier Interfaces
Fluidic channels lined with endothelial cells allow us to introduce vascular-like interfaces that tissues exchange nutrients/excretory/metabolites. This forces cells to communicate across these engineered biological barriers (Blood-Brain Barrier, Gut-Blood Barrier, etc.).
Low-Absorption Thermoplastic Fabrication:
Addressing the industry-wide challenge of PDMS drug absorption, we utilize medical-grade thermoplastics (e.g., COC, COP). This ensures chemical stability and prevents the non-specific binding of small-molecule drugs, providing unparalleled accuracy for PK/PD modeling.
Integrated Sensing Layer
Non-invasive, embedded biosensors continuously monitor key physiological parameters, including oxygen tension, pH gradients, and transepithelial electrical resistance (TEER). These real-time readouts provide continuous insight into tissue health, barrier integrity, and system stability throughout long-term experiments.
Physiological Integration and System Scaling
Physiological Scaling: We utilize allometric scaling and PBPK (Physiologically Based Pharmacokinetic) modeling to calculate the relative volumes and flow rates of each organ compartment, ensuring the residence time of media matches human blood flow proportions.
Universal Blood Surrogate: We have developed serum-free, common culture media that support the simultaneous health of diverse cell types without compromising individual phenotypes.
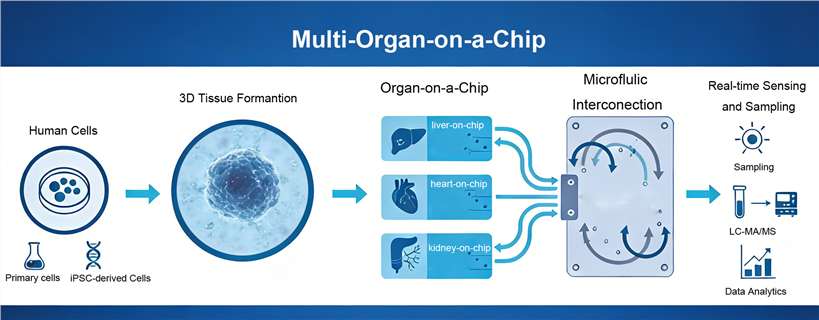
Applications in Drug Discovery and Translational Research

Systemic ADMET Profiling
Predicting how a drug is metabolized in the liver before reaching the heart or kidneys.

Cascade Toxicity Research
Studying how metabolites generated in one organ trigger secondary injury in distal tissues.

Immune-Tumor Interactions
Modeling the circulation of immune cells and their infiltration into tumor niches.

Environmental Toxicology
Assessing the systemic impact of inhaled pollutants as they move across the lung-blood barrier.
Reference
- Chen C, Zhou L, et al. From organ to system: Multi-organ-on-a-chip platforms as next-generation biomedical simulators. Biofabrication. 2025 Oct 21;17(4).
